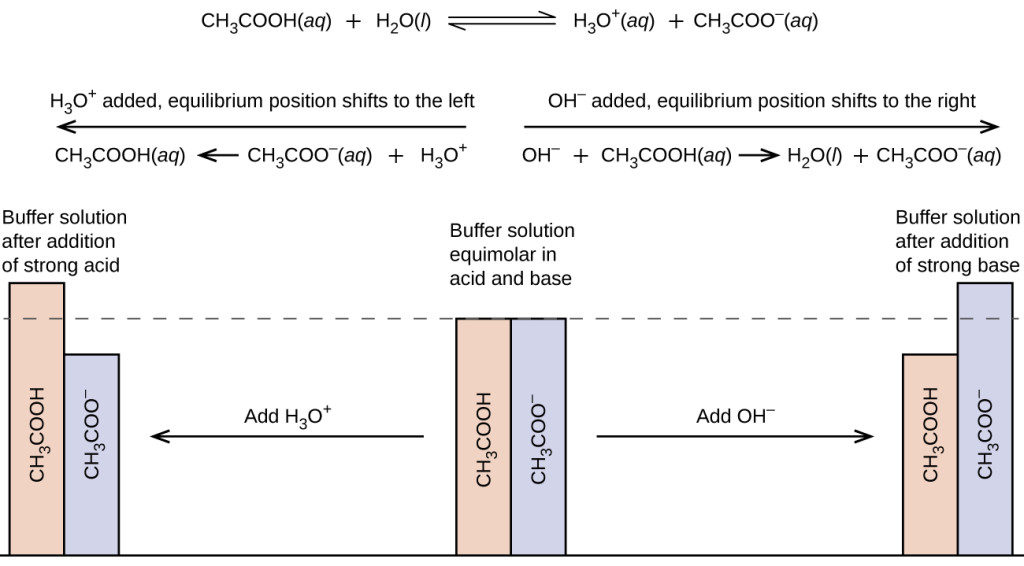
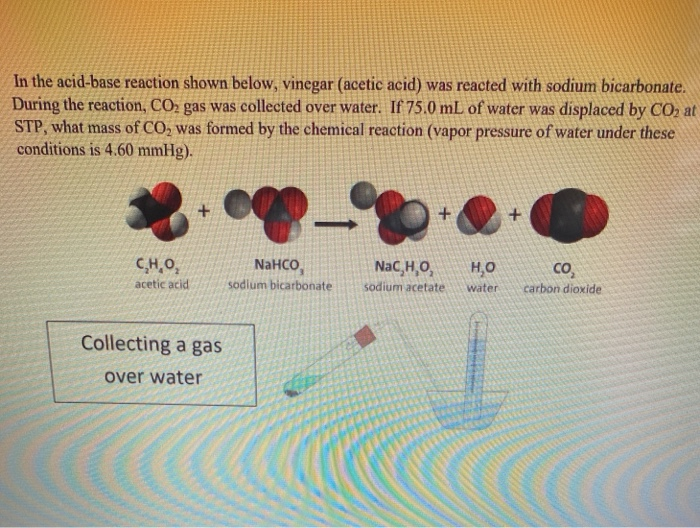
SOLVED: Sodium acetate (NaC2H3O2) is a basic salt. When sodium acetate is dissolved in water, it dissociates into its component ions. This reaction goes to completion, as indicated by the one-way arrow
Prove the buffer action of acetic acid and sodium acetate by the addition of 0.01 mol of solid sodium hydroxide. - Sarthaks eConnect | Largest Online Education Community

Two buffer solutions, `A` and `B`, each made acetic acid and sodium acetate differ in their `pH`... - YouTube
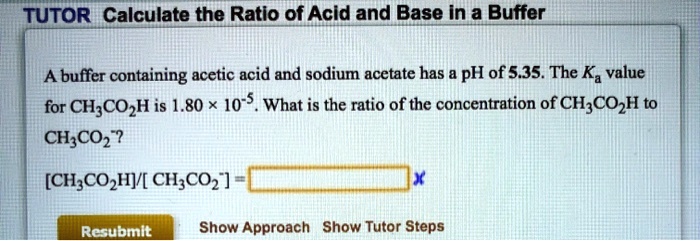
SOLVED: TUTOR Calculate the Ratio of Acid and Base In a Buffer A buffer containing acetic acid and sodium acetate has pH of 5.35. The Ka value for CH;COzH is 1.80 x

SOLVED: 16. Which of the following could be added to a solution of sodium acetate to produce a buffer? A) acetic acid only B) acetic acid or hydrochloric acid C) hydrochloric acid

SCH 4 U 1. What are buffers? Buffers are mixtures of conjugate acid- base pairs that allow a solution to resist changes in pH when acids and/or bases. - ppt download

Sodium Acetate(CH3COONa) - Structure, Properties, Preparations, Uses, Important questions, FAQs of sodium acetate.

50 mL of 0.1 M solution of sodium acetate and 50 mL of 0.01 M acetic acid are mixed. The pKa of acetic acid is 4.76. The pH of the buffer solution is:

In a mixture of acetic acid and sodium acetate, the ratio of concentrations of the salt to the acid is increased ten times. Then the pH of the solution:







![BS005] 3M Sodium Acetate, pH 5.2 | Biosolution BS005] 3M Sodium Acetate, pH 5.2 | Biosolution](http://biosolution.cafe24.com/wp-content/uploads/2016/02/BS005-Sodium-Acetate-Solution.jpg)